The GLP-1 Ripple Effect: How U.S. Compounding Is Transforming (2006–2026)
Compounding in the U.S. looks very different today than it did 20 years ago. What was once a fragmented landscape of primarily 503A pharmacies under comparatively less intensive and more variable regulatory oversight has evolved into a more complex, highly regulated ecosystem that includes both 503A compounding pharmacies and 503B outsourcing facilities.
In recent years, the rise of GLP-1 agonists has brought unprecedented attention to the space. But GLP-1s alone don’t explain the transformation. These drugs acted as a catalyst, accelerating visibility into shifts that were already underway.
We explore the key events and forces that have shaped compounding in the U.S. today, from regulatory shifts and drug shortages to changing business models, market dynamics, and public perception.
U.S. Compounding 20 Years Ago vs Today
Compounding 20 Years Ago (Pre-2012 Snapshot)
About 20 years ago, Section 503B outsourcing facilities did not yet exist, and the compounding landscape was primarily composed of 503A pharmacies. These operations spanned hospital systems, infusion pharmacies, and "mom-and-pop" community pharmacies.
At the time, pharmacies primarily prepared Compounded Sterile Preparations (CSPs) and Compounded Nonsterile Preparations (CNSPs), relying largely on manual, legacy methods. Offerings were broad, with many sites maintaining extensive formularies that could include hundreds to thousands of formulations.
Production was typically small in scale, with batch sizes often serving just one to ten patients. Business models reflected a mix of cash-pay and insurance-based reimbursement.
Regulatory oversight was comparatively more limited and varied across jurisdictions. Board certification had not yet been established, and the level of scrutiny and enforcement was significantly lower than what the industry experiences today.
Yet, this was a critical period preceding the transition of compounding from a traditionally accepted, localized practice to a growing component of specialized pharmaceutical care. Pharmacy compounding began re-emerging as a critical, yet increasingly scrutinized, area of practice, with increasing concerns regarding quality control and regulatory oversight.
At the same time, underlying pressures were beginning to surface. Demand for compounded sterile preparations was increasing, particularly in response to hospital drug shortages. Some pharmacies began operating at a larger scale, including interstate dispensing.
As this shift took hold, concerns emerged around quality, consistency, and regulatory oversight. Tensions reached a turning point, setting the stage for the regulatory and structural changes that would redefine compounding in the years that followed.
Compounding Today (2026 Snapshot)
Today, the compounding landscape consists of a mix of 503A pharmacies and 503B outsourcing facilities. Collaboration has become more strategic, with 503B outsourcing facilities increasingly supplying 503A pharmacies, subject to applicable state and federal requirements. While these operations still span hospital systems, infusion providers, and community pharmacies, ownership models have become far more diverse. These range from independent and health system–aligned entities to VC- and private equity–backed organizations, as well as telehealth-integrated platforms.
Compounding practices have also evolved. While sterile-to-sterile CSPs remain common, there has been an observed increase in nonsterile-to-sterile Compounded Sterile Preparations (CSPs) and Compounded Nonsterile Preparations (CNSPs), reflecting both changing demand and advances in capability.
Meanwhile, operational processes have begun to diverge. Some facilities continue to rely on manual, legacy methods, while others have adopted semi-automated or fully automated (robotic) compounding systems to improve compliance and scalability.
Another key shift is in the structure of offerings. Many compounding pharmacies now maintain more focused formularies, preparing fewer formulations overall of intentional batch sizes for particular patient cohorts.
Business models have followed suit, with cash-pay arrangements becoming increasingly predominant, particularly in certain direct-to-patient and telehealth-driven settings.
Professionalization has also advanced. The Board of Pharmacy Specialties now offers the Board Certified Sterile Compounding Pharmacist (BCSCP) credential, recognizing advanced expertise in sterile compounding practices, quality systems, and compliance with minimum industry standards (e.g., USP <797>).
These operational and structural changes have unfolded alongside significant regulatory evolution. Both 503A and 503B facilities now operate under heightened scrutiny, with increased expectations around quality, documentation, and enforcement across the compounding sector.
What has emerged in 2026 is a compounding landscape that’s more structured, segmented, collaborative, and visible in healthcare conversations—but one that is facing increasing pressures, challenges, and expectations that will need to be met with a strategic industry mindset.
Founded in 2019 at a pivotal moment for the compounding industry, Restore Health Consulting has evolved alongside its transformation. As the landscape expanded beyond 503A and 503B into GMP, telehealth, med spas, QC labs, and cell and gene therapy, so did the need for more integrated and strategic support. Today, we help healthcare and pharmaceutical organizations navigate complexity, strengthen compliance, and operate with confidence in a rapidly evolving environment.
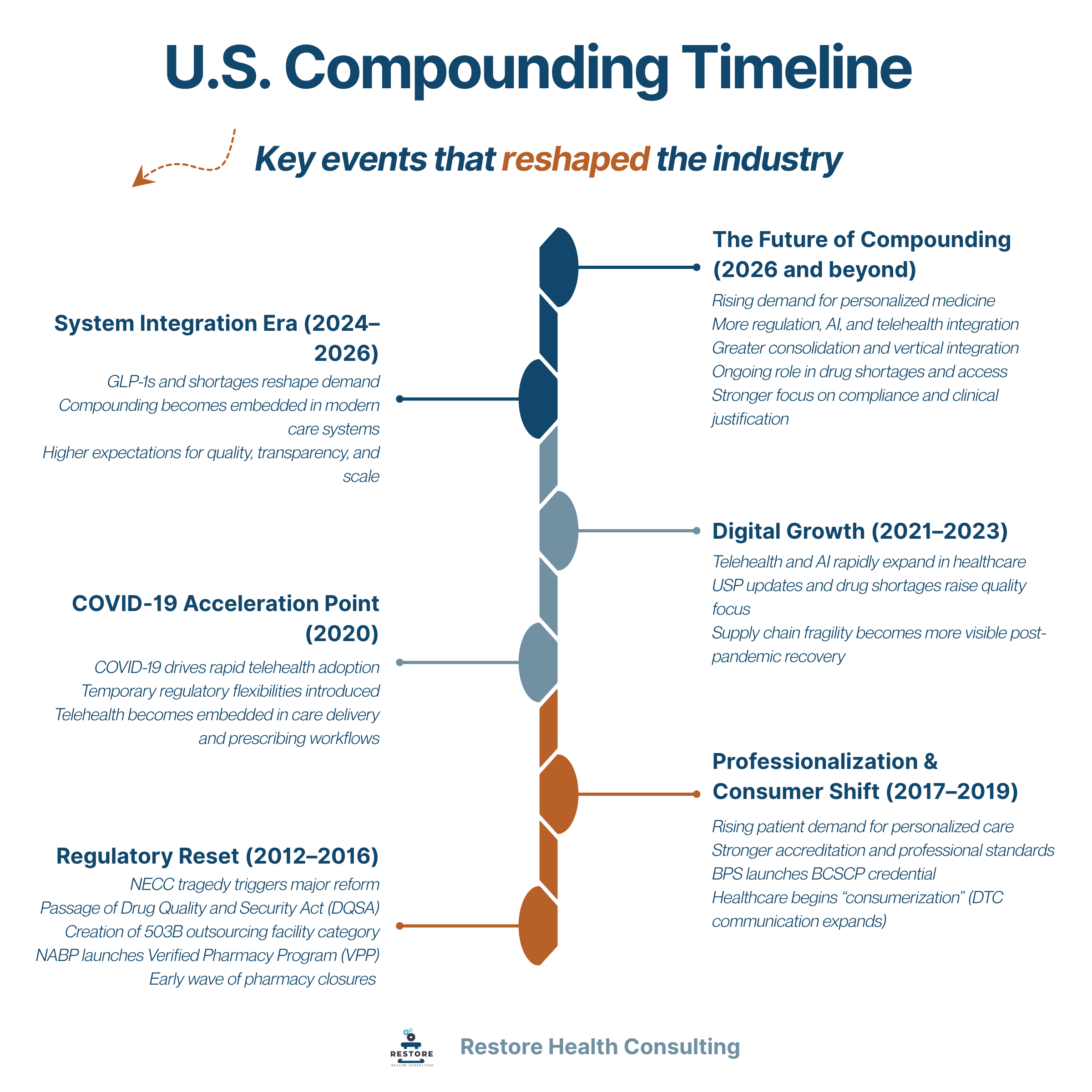
Key Shifts That Reshaped the Industry (2012–2026)
The modern compounding landscape did not change overnight. It was shaped by a series of defining events, regulatory responses, and cultural shifts that collectively redefined how the industry operates today.
What began as a response to safety failures has evolved into a broader transformation—one that includes tighter oversight, rising patient expectations, new care delivery models, and accelerating technological change. The timeline below highlights the key inflection points that moved compounding from a localized practice to a more structured, visible, and strategically important part of healthcare.
NECC Tragedy and the Birth of 503B Outsourcing Facilities
The modern compounding landscape began to take shape in 2012 following the New England Compounding Center (NECC) tragedy, a multistate fungal meningitis outbreak linked to contaminated injectable steroid medications that resulted in over 50 deaths. The event was followed by significant regulatory response and led to the passage of the Drug Quality and Security Act (DQSA), which amended the FD&C Act and formally established the 503B outsourcing facility category.
This marked a structural shift in U.S. compounding. 503B outsourcing facilities were created to provide a regulated pathway for larger-scale sterile compounding, under strengthened FDA oversight and enhanced quality expectations, particularly for products not readily available from commercial manufacturers.
Tightening Regulations and Accreditation
In 2013, state Boards of Pharmacy began to tighten compounding regulations, increase frequency of inspections, and resulted in expanded expectations for quality and compliance.
The National Association of Boards of Pharmacy (NABP) introduced the Verified Pharmacy Program (VPP) in 2013. The program aimed to support more consistent inspection standards for non-resident and compounding pharmacies. Over time, VPP has become a widely recognized accreditation pathway, helping state boards evaluate compliance with state regulations and USP standards, including USP <795>, <797>, and <800>. By 2023, the program had completed more than 2,400 on-site inspections.
As regulatory infrastructure was evolving, broader cultural and behavioral shifts were also taking place. Beginning in the mid-2010s, healthcare professionals increasingly became visible voices in digital and social media spaces, contributing to the growing consumer awareness of personalized and precision health approaches. Patient expectations began shifting toward more individualized care models and a greater focus on wellness and longevity.
By 2016, several compounding pharmacies had closed due to the increase in costs to become compliant or from regulatory enforcement actions or operational closures. 503B outsourcing facilities began cropping up, and regulations for both entities began to tighten further.
Healthcare in Social Media and Professionalization
By 2019, healthcare marketing had fully entered mainstream social media platforms, with consumer-facing campaigns appearing on channels such as TikTok. This reflected a broader trend toward the consumerization of healthcare communication and increased direct engagement between healthcare brands and patients.
That same year, the Board of Pharmacy Specialties (BPS) launched the Board Certified Sterile Compounding Pharmacist (BCSCP) credential, formalizing advanced specialization in sterile compounding practice. The initial cohort of pharmacists certified in 2019 will reach their first recertification cycle in 2026, marking an important milestone in the professionalization of the field.
The COVID-19 Pandemic, Drug Shortages, GLP-1s, and Telehealth
In 2020, the COVID-19 pandemic accelerated the adoption of telehealth and remote care models, rapidly shifting care delivery into virtual environments. During this period, certain regulatory flexibilities were introduced to support continuity of care and access.
While COVID-19 made telehealth mainstream, it continued to solidify as a major part of healthcare post-pandemic. Telehealth has now become integrated into prescribing workflows, pharmacy operations, and patient access models, including pathways that influence compounding utilization.
Following the pandemic, from 2022 to 2025, multiple national shortages of GLP-1 receptor agonists placed additional pressure on both 503A and 503B compounders, who played a role in supporting continuity of care during supply constraints.
Although GLP-1 therapies first emerged in 2005 with the approval of exenatide (Byetta), their clinical and commercial prominence expanded significantly in the 2020s. Key milestones included the approval of liraglutide (Saxenda) in 2014 for weight management, semaglutide (Ozempic) in 2017 for diabetes management, and Wegovy in 2021 as a higher-dose formulation for obesity treatment, followed by continued development of higher-dose oral semaglutide formulations being studied for weight management. As of this writing, oral semaglutide (Rybelsus) is approved in the U.S. for type 2 diabetes, and weight management indications for oral formulations have not yet received FDA approval.
AI, Innovation, and Supply Chain Weaknesses
Advances in AI beginning around 2022 began influencing multiple areas of healthcare, including drug development, operations, and patient engagement. In 2026, AI is deeply embedded across healthcare and compounding, often supporting formulation design, quality systems, operations, and patient engagement. At the same time, it introduces new challenges around validation, data integrity, regulatory interpretation, and responsible use, requiring organizations to balance innovation with strong oversight and quality control.
In 2023, updates to USP standards further increased quality expectations, with major revisions to chapters <795> (non-sterile), <797> (sterile), and <800> (hazardous drugs). The update focused on enhanced safety, clearer Beyond-Use Dates (BUDs), and stringent personnel training.
In 2025, persistent drug shortages underscored deeper structural weaknesses in global supply chains, bringing compounding into focus as a critical solution for maintaining patient access. At the same time, the industry continued to mature, with stronger emphasis on quality systems, governance, and collaboration, particularly within the 503B sector. As costs rose and regulatory engagement increased, compounding became more clearly integrated into the broader healthcare system, reflecting both its growing importance and the pressures shaping its future.
The new U.S. administration maintained regulatory continuity with the FDA being involved in targeted initiatives, including the Green List for GLP-1s, HRT black box removal, and increased warning letters on claims in drug marketing (particularly by telehealth firms).
The GLP-1 Ripple Effect on Compounding in the U.S.
Why GLP-1s Changed Visibility, Not Just Volume
GLP-1 therapies have become one of the most visible developments in modern pharmaceutical care, not only because of clinical use, but because of widespread public and commercial attention. They’ve driven lawsuits, media coverage, and public debate on a scale not seen in years.
From an operational perspective, GLP-1 demand has increased compounding workload. According to APC 2025–2026 survey data, GLP-1 preparations ranked among the top three compounded therapies by volume. In response, approximately half of respondents expanded staffing or operating hours and invested in equipment, while about one quarter expanded facilities or moved to larger sites.
However, the impact of GLP-1s extends beyond volume. These therapies have gained rapid visibility due to media coverage, social media, celebrity influence, and sustained public discussion around weight management and metabolic health. As a result, GLP-1s have become a high-profile category within both clinical practice and consumer health conversations, increasing attention on compounding as a delivery channel.
This visibility has also led to increased regulatory scrutiny. The 2022–2025 national drug shortages drove expanded compounding activity, followed by closer review from regulators as supply conditions stabilized. In this context, compounding continues to be governed by statutory and regulatory frameworks that generally limit its use to circumstances involving patient-specific needs or other conditions described under Sections 503A and 503B.
Despite the increased attention compounding got, public understanding remains limited. A 2025 national poll found that:
42% of U.S. adults are familiar with pharmacy compounding
Nearly one-third do not understand what compounding is or what it involves
79% patients who used compounding pharmacies report a positive experience
This reflects increasing awareness of compounding, but continued gaps in understanding of its regulation and oversight. Continued focus on quality, compliance, and transparency will be key to strengthening trust and establishing its role within modern healthcare.
The Response of Major Pharmaceutical Manufacturers
As GLP-1 therapies gained clinical and commercial prominence, they also attracted increased attention from large pharmaceutical manufacturers. The response has taken two parallel directions: protecting existing market positions and repositioning to integrate with compounders as personalized medicine expands.
1. Defensive Response: Litigation and Messaging
One response has been increased legal and regulatory activity related to GLP-1 product categories. This has involved litigation involving compounded versions of GLP-1 medications and telehealth platforms, as well as expanded scrutiny of telehealth platforms and compounding practices.
Manufacturers have also strengthened public messaging campaigns emphasizing distinctions between FDA-approved products and compounded medications.
2. Strategic Response: Expansion Into Personalized and Specialty Markets
At the same time, generic and specialty pharma companies are looking for new revenue streams. Some healthcare entities and drug manufacturers are increasingly recognizing the strategic importance of compounding in helping meet unique patient needs and personalized medicine demand.
In turn, another response from healthcare/pharma and telehealth companies has been to invest into 503A and/or 503B facilities, often as part of vertical integration. This strategy strengthens control of the supply chain and allows companies to capture drug product margins and bypass traditional pharmacy benefit managers (PBMs).
Multiple industry reports forecast continued growth in the compounding pharmacy market, projecting expansion from approximately USD 14.7 billion in 2025 to USD 19.4 billion by 2030, representing a compound annual growth rate (CAGR) of around 5.7%. This growth is closely linked to demand for personalized medicine, which is increasingly viewed as a strategic area of long-term value creation.
Within this shift, it’s evident that compounding pharmacies play a critical role by supporting patient-specific medication needs that are not met by commercially available products. This reinforces their position within the broader personalized medicine ecosystem.
Specialty drugs and pharmacy services continue to grow rapidly and attract investment from large healthcare/pharma sectors. Compounding and personalized dosing are recognized as integral to new pharmaceutical business models. Indeed, industry analysis shows high specialist demand and investment in specialty medicines, including drug distributor Cardinal Health raising forecasts due to specialty demand.
As a result, compounding and individualized dosing are becoming integrated into the wider specialty care infrastructure rather than viewed as a standalone or niche function.
Overall, the GLP-1 era has accelerated a dual shift in the industry, where compounding is simultaneously more scrutinized and more strategically embedded in healthcare delivery. As manufacturers, providers, and investors adjust to this reality, compounding is increasingly being defined not by its fringes, but by its role in enabling personalized, accessible, and evolving models of care.
Regulatory Scrutiny Intensified
Regulatory oversight of compounding pharmacies in the GLP-1 era has become more structured and targeted, with several key developments shaping enforcement and inspection frameworks across the U.S.
State boards of pharmacy have increasingly relied on the National Association of Boards of Pharmacy (NABP) Verified Pharmacy Program (VPP) to improve consistency in inspections and support shared oversight models for non-resident and compounding pharmacies. As adoption has expanded, VPP has become a more widely used mechanism for standardizing how compliance with USP <795>, <797>, and <800> is assessed across jurisdictions.
At the federal level, the U.S. Food and Drug Administration introduced the “Green List” import alert (66-80), sometimes referred to as a “green list,” as a regulatory tool intended to strengthen oversight of active pharmaceutical ingredient (API) sourcing for GLP-1 drugs. The initiative is intended to facilitate entry of APIs from manufacturers that have undergone FDA inspection while detaining those from firms that have not been verified.
In practice, however, identifying specific API manufacturers that meet these criteria may not be straightforward, as publicly available information regarding qualifying sources is limited and may not be presented in a centralized or easily interpretable format.
While the FDA has framed this approach as a patient safety measure, its practical application may present challenges for compounders attempting to independently verify API sourcing against regulatory expectations, and some pharmacists, compounding advocates, and industry observers have expressed skepticism regarding its operational impact.
In parallel, the FDA has issued multiple warning letters and enforcement actions related to promotional claims involving compounded as well as FDA-approved drug products. As of early 2026, FDA communications reflect increased enforcement activity targeting false or misleading promotional practices, including claims that may inappropriately equate compounded products with FDA-approved drugs or otherwise omit or minimize risk information.
These actions have involved a range of entities, including pharmaceutical manufacturers, telehealth platforms, and compounding-related operations.
These actions reflect increased attention on how both compounded and commercially manufactured medications are marketed and communicated, particularly in categories experiencing high public and commercial visibility.
Together, these developments reflect a more coordinated and evolving regulatory approach focused on inspection standardization, supply chain control, and enforcement of promotional compliance within compounding-related activities.
Business Model Shifts
In the post-GLP-1 and post-USP update environment, compounding pharmacies have been reassessing how their product portfolios and operations are structured. One clear shift has been the rationalization of SKU lists, with some organizations moving away from historically larger formularies.
This change reflects both regulatory pressure and operational reality. As oversight expectations and documentation standards have increased, maintaining very large, highly customized product catalogs has become more difficult to sustain at scale.
In response, some compounders are shifting toward more focused portfolios built around higher-frequency, significantly different preparations. The emphasis is moving toward high-demand products and streamlined operations rather than broad customization across large numbers of formulations.
As a result, facilities now have fewer products but with enhanced focus on safety and quality control. The move is toward greater standardization and high-quality production over endless customization. This shift also reflects a broader maturation of the sector, where compounding is pivoting from a "make anything" model to a specialized, high-compliance model, prioritizing compliance, quality, and operational sustainability.
Current U.S. Compounding Market Dynamics
Market Expansion and New Entrants
The GLP-1 shortage drew non-traditional players into compounding, including venture capital firms, private equity investors, telehealth platforms, and entrepreneurs.
As a result, compounding has undergone consolidation and bifurcation. There seem to be fewer “mom-and-pop” shops and more large-scale compounders with professional infrastructure are emerging.
Two new archetypes emerged within the market:
Speed/marketing-led entrants optimizing for rapid growth
Quality-focused entrants building for long-term scale or acquisition
Overall, there has been increased investment in quality systems and compliance infrastructure among certain operators as serious players invested in compliance and sustainability.
503A and 503B Collaboration
The 503A/503B framework allows compounding facilities to complement each other:
503As deliver patient-specific personalization and formulation adaptation
503Bs provide standardized compounds in sufficient quantities to meet predictable demand and mitigate shortages
503As may purchase 503B-prepared compounds for individual patient prescriptions (subject to state and federal regulations), extending the range of treatment options
Together, these facilities contribute to the operational infrastructure that supports individualized approaches to medication prescribing.
Changing Patient Awareness and Behavior
Patient behavior is shifting toward more direct, individualized engagement with healthcare, with a growing segment willing to pay out of pocket for personalized treatments rather than relying solely on traditional insurance-based models. This includes increased interest in therapies tailored to individual genetics, health status, and lifestyle factors.
Out-of-pocket healthcare spending continues to rise, reaching approximately $556.6 billion in 2024, representing 11% of total healthcare expenditure. This trend is driven in part by the growth of high-deductible health plans, along with more patients seeking care outside traditional insurance networks.
These shifts are particularly pronounced among younger populations. Gen Z and Millennials are more likely to engage with healthcare through digital and retail channels, with 79% reporting regular use of tools such as wearables, telehealth, or online prescription services, and approximately 40–42% using virtual visits within the past year.
This shift in engagement is changing how patients access compounded therapies. Rather than relying solely on traditional referral pathways, patients are increasingly entering the system through telehealth platforms, direct-to-consumer models, and digital health platforms. In these settings, expectations around convenience, speed, and personalization are higher, and treatment decisions are often more collaborative or patient-informed.
For compounding pharmacies, this represents both an opportunity and a constraint. Demand is increasingly shaped by patient awareness and preference, particularly in high-visibility categories such as metabolic health, hormonal health, and wellness-oriented services. At the same time, these channels bring greater scrutiny around prescribing appropriateness, claims, and regulatory compliance.
As a result, patient behavior is no longer a downstream factor—it is increasingly influencing how compounded therapies are accessed, delivered, and evaluated within the broader healthcare system.
Where is Compounding Headed in 2026 & Beyond?
1. Demand for Personalized Medicine is Expected to Continue
Demand for personalized medicine is expected to continue, with patients seeking convenient access to individualized treatments. As expectations rise, greater attention is being placed on the source and quality of ingredients used in compounding. Novel equipment can level up the reproducibility and reliability of the compounding process, supporting scaling and regulatory compliance.
In response, compounders may need to take a barbell approach to formulation offerings—balancing precision medicine offerings with popular stock-keeping unit (SKU) expectations.
2. Peptides Will Keep Drawing Attention
Regulatory guidance around certain peptide ingredients is still evolving, and compounders are monitoring how access pathways may take shape. Any future framework will likely come with stricter expectations around quality and control. This creates an opportunity for the industry to reinforce discipline, transparency, and a strong commitment to high standards.
3. Regulations Will Continue to Evolve as AI and Telehealth Expand
While federal courts provided some clarity in 2025, regulatory activity is increasingly shifting to the state level. Early state-level legislative activity, such as Florida Senate Bill 860, illustrates the direction of potential regulatory focus. Although not enacted, the bill proposed additional requirements around API sourcing, documentation, and quality verification for compounded weight-loss medications, reflecting increasing attention to compounding at the state level.
This evolving landscape creates opportunities for the industry to engage earlier, strengthen education, and reinforce its role in expanding safe and responsible patient access through compounding.
As telehealth and AI continue to expand, regulatory frameworks and industry standards are expected to evolve alongside them. Compounding pharmacies will be required to operate with greater consistency, including predictable turnaround times, structured documentation, and high levels of traceability across the compounding process.
These models also place greater emphasis on clear, auditable communication between prescribers and pharmacies, as well as more standardized, quality-driven operations aligned with GMP-level expectations.
In response, compounding platforms are evolving to support these workflows, including prescriber portals, order tracking systems, and integrated electronic communication. Pharmacy management systems are also adapting to accommodate higher volumes of telehealth-driven prescribing and more coordinated care delivery.
4. Vertical Integration Will Reshape Healthcare
Vertical integration—the consolidation of multiple parts of the healthcare supply chain under a single enterprise—is expected to play a defining role in the future of compounding. Pharmacies, telehealth platforms, laboratories, and specialty services are increasingly being combined into unified operating models, shifting value from standalone dispensing toward ownership of the patient relationship and care pathway.
This trend is already visible in direct-to-consumer telehealth platforms and private equity-backed groups, which are building integrated systems that may include prescribing, telehealth access, 503A pharmacies, and in some cases 503B outsourcing facilities, internal quality control labs, and API sourcing capabilities. The result is the emergence of multi-entity healthcare platforms that operate across several layers of care delivery rather than within a single segment.
Telehealth companies have often led this shift by building or acquiring pharmacy capabilities. Increasingly, however, compounding pharmacies may pursue integration in the opposite direction—adding telehealth interfaces and adjacent services to extend patient access and strengthen their role within the care continuum. This evolution suggests a move beyond traditional 503A operations toward more integrated delivery models that are also attractive for acquisition.
5. Drug Shortages May Still Be Expected
Drug shortages are likely to persist even if they receive less public attention in 2026 and beyond. Underlying pressures such as global supply constraints and cost dynamics continue to challenge the availability of older, low-margin generic medicines, which may further limit manufacturing participation in the U.S. market. In this context, compounders remain an important part of the healthcare system, helping maintain continuity of care when traditional supply chains fall short.
6. Claims & Clinical Justification Will Continue to Be Critical
As regulatory scrutiny continues, limitations around claims remain a central consideration in compounding. Compounded medications are generally not permitted to be promoted with claims of safety or efficacy, reinforcing the need for careful, compliant communication across all channels.
At the same time, justification for the use of a compounded preparation over an FDA-approved product remains an important consideration. Prescribing decisions may be supported by documentation reflecting patient-specific needs, consistent with established compounding frameworks and regulatory expectations.
Key Takeaways
Taken together, these trends show a compounding industry that is becoming more structured, more integrated, and more visible in healthcare. What was once a highly localized, flexible practice is now operating with clearer standards and expectations. At its core, compounding is being shaped by both rising external pressures and its continued role in enabling personalized patient care.
Work with Restore Health Consulting to navigate today’s evolving compounding landscape with clarity and confidence.
Disclaimer: This article is intended to provide general information on U.S. compounding and FDA actions related to GLP-1 compounding and supply chain oversight. It should not be construed as legal, regulatory, or medical advice. Readers are encouraged to consult an attorney for guidance specific to their circumstances.