Med Spa Compliance: Key Trends, Risks & Guidance for Owners
Medical spas operate at the intersection of medicine, aesthetics, and business, which makes regulatory compliance uniquely challenging. While med spas provide non-surgical cosmetic procedures, their offerings often involve prescription drugs, energy-based devices, and invasive interventions, each carrying legal and clinical risk. Creating a compliance-ready med spa typically involves structured oversight, well-documented policies, and ongoing risk management.
Note: This article is intended for educational purposes and does not constitute legal advice.
The Fast Growth of Med Spas in the U.S.
Medical spas or “med spas” have emerged as one of the fastest-growing segments of outpatient healthcare in the United States. These hybrid businesses combine traditional spa services with medical treatments such as injectable aesthetics, energy-based devices, intravenous hydration therapy, and weight management programs.
Growth is driven by demand for:
Minimally invasive cosmetic procedures
Injectable aesthetic treatments
IV hydration therapy and wellness services
Hormone and metabolic therapies
Weight management medications
As demand for these services grows, med spas are increasingly functioning as clinical environments where prescription medications are stored, administered, dispensed, or otherwise used in connection with patient care.
If you need a compliance partner for your med spa, Restore Health Consulting provides support for licensure, medication sourcing and storage, and operational compliance. Book a free med spa compliance intro call with us to get started.
Fragmented Regulatory Oversight
Despite the clinical nature of many services, regulatory oversight of medical spas remains fragmented. Unlike hospitals, physician practices, or pharmacies, med spas often operate in a regulatory area where multiple professional boards (medical, nursing, pharmacy, and cosmetology) share responsibility but no single framework governs the entire operation.
Medical spas now operate in a wide variety of environments, including:
Dedicated wellness clinics
Dermatology or plastic surgery practices
Luxury hotels and resorts
Retail shopping centers
Fitness and lifestyle facilities
This rapid expansion has transformed many med spas into healthcare delivery environments where prescription medications and sterile injectable products are routinely administered.
However, regulatory oversight has not evolved at the same pace. For this reason, med spa compliance remains a growing regulatory challenge that needs to be carefully considered. Because medical spas operate across medical, nursing, pharmacy, and cosmetic regulatory frameworks, operators often must navigate multiple regulatory regimes simultaneously.
For owners and operators, building a structured compliance program is essential to ensuring regulatory alignment, patient safety, and long-term business sustainability. It’s important to consider differences across states, ownership models, scopes of practice, and overall regulatory requirements.
Regulatory Challenges Facing Med Spas
Medical spas present a unique regulatory challenge because they combine aspects of healthcare delivery with retail wellness services. Multiple regulators may have jurisdiction, including:
State medical boards
Boards of nursing
Boards of pharmacy
Cosmetology boards
Local health departments
The result is a patchwork regulatory structure where requirements vary significantly across states.
In many jurisdictions, med spas operate without a dedicated facility license. Instead, regulatory oversight focuses primarily on the healthcare professionals delivering services.
In addition, med spas must carefully source FDA-approved and/or compounded (when appropriate) medications, verify their quality and compliance, and maintain proper storage conditions to support medication integrity and patient safety.
Med Spa Compliance Program: Key Considerations
Operating a compliant medical spa requires a structured regulatory and operational framework. Because most med spa treatments are considered the practice of medicine in many jurisdictions, operators must ensure their facility and staff follow all applicable legal, safety, and ethical standards.
A strong Med Spa Compliance program should address:
Staff credentials and licensing: All providers, including physicians, nurse practitioners, physician assistants, and RNs, must hold current licenses and meet state-specific scope-of-practice requirements.
Ownership and corporate structure: Facility ownership often influences regulatory responsibilities. Physicians typically must retain medical control, while non-physician owners may participate via Management Services Organizations (MSOs).
Patient safety and clinical protocols: Implementing standardized protocols, delegation guidelines, and emergency procedures protects patients and ensures treatments meet the standard of care.
Privacy and data security: Compliance with HIPAA and state patient privacy laws is essential for safeguarding health information.
Marketing and advertising: Claims must be truthful, non-misleading, and compliant with medical advertising rules.
A robust compliance program ensures a med spa operates legally, reduces liability, maintains patient trust, and prepares the facility for inspections. In essence, Med Spa Compliance is about building safe, accountable, and professional practices that protect both patients and your business.
Facility Licensing and Registration for Med Spa Compliance
In most states, med spa facilities are not required to obtain a dedicated medical spa license or registration. Instead, regulation typically applies to health care professionals who deliver the services, including:
Physicians
Nurse practitioners (NPs)
Physician assistants (PAs)
Registered nurses (RNs)
However, some states have implemented additional facility-level oversight.
Ohio’s model is particularly notable. Any entity that possesses or distributes prescription medications must register with the Ohio Board of Pharmacy as a terminal distributor.
This designation requires compliance with rules governing:
Drug procurement
Storage
Inventory control
Administration and documentation
Because the definition of “dangerous drugs” includes all prescription medications, many med spas offering injectable treatments fall under pharmacy regulatory oversight.
The lack of consistent facility-level regulation means patients may receive prescription drug treatments in environments that operate under different regulatory frameworks than hospitals or pharmacies.
Services and Treatments Offered in Medical Spas
Medical spas typically offer a range of aesthetic and wellness services, including:
Injectable Aesthetic Treatments
Injectable products generally fall into two categories.
Dermal Fillers
Common products include:
Juvederm
Restylane
These FDA-approved hyaluronic acid fillers are used to restore facial volume and contour.
Other popular options include Radiesse (calcium hydroxylapatite) for collagen stimulation, Sculptra (poly-L-lactic acid) for structural support, and Belotero or Revanesse for fine lines.
Certain peptides are also being used as dermal fillers in medical spas. However, several safety and regulatory concerns have been raised, especially in regards to the uncertainties around peptide compounding, sourcing, and quality.
In September 2023, the FDA updated the list of bulk drug substances nominated for use under Section 503A. Several peptides were added to Category 2 because FDA claimed to identify significant safety risks. This move has since been followed by additional FDA updates and policy signals, including the September 2024 interim bulks list update.
Neurotoxins
Examples include:
Botox
Dysport
Xeomin
These products temporarily reduce muscle activity responsible for wrinkles.
In addition, Argireline (another peptide) is often marketed as a "topical Botox" alternative. However, argireline is not a bacterial neurotoxin. Argireline is also not FDA-approved for use as a drug because it is classified as a cosmetic ingredient.
Energy-Based Devices
Energy-based procedures include:
Laser resurfacing
Intense Pulsed Light (IPL) therapy
Radiofrequency skin tightening
Cryolipolysis (fat freezing)
Lasers often represent one of the highest-margin services in the medical spa industry.
IV Hydration Therapy
IV therapy has become increasingly common in med spas and wellness clinics. These treatments typically involve intravenous administration of fluids, vitamins, minerals, and electrolytes.
However, IV therapy may introduce clinical risks associated with:
Sterile preparation of injectable products
Contamination
Dosing errors
Adverse reactions
Preparing IV mixtures may also constitute sterile compounding, depending on how the products are prepared.
Because of these risks, many states have issued guidance or regulations addressing IV therapy practices.
For example:
South Carolina regulators have clarified that IV therapy involves the practice of medicine, nursing, and pharmacy simultaneously.
California’s Board of Pharmacy has indicated that preparing IV mixtures may constitute sterile compounding, which may require preparation in compliant environments with appropriately trained personnel.
To ensure compliance at your medical spa, it’s suggested to put these protocols in place for your IV treatments:
Abide by your state’s CPOM and scope of practice laws
Establish a patient-provider relationship through a Good Faith Exam.
Ensure a provider determines if a patient qualifies for treatment
Practice safe and compliant compounding under trained personnel
Have standardized operating procedures (SOPs) for IV administration
Peptides
Although peptides are seeing a boom in med spas and wellness space, the regulatory status of peptide compounding remains a debated issue.
As of 2023, the FDA has significantly restricted which peptides can be legally compounded. Peptides with 40 or fewer amino acids may be eligible for compounding only if they meet applicable requirements under Section 503A, such as appearing on the FDA’s 503A Bulks List, having a USP monograph, or being components of FDA-approved drugs.
Peptides with more than 40 amino acids are generally treated as biologics and typically fall outside traditional pharmacy compounding pathways.
Certain peptides marketed in the wellness industry may not meet current FDA criteria for compounding.
Many peptides are classified under Category 2, indicating that FDA has identified safety concerns and that these substances generally should not be compounded until and unless they are moved to Category 1 of the FDA’s interim bulks list or the final FDA bulks list. These include: BPC-157, Thymosin α1, Thymosin Beta-4 Fragment, CJC-1295, Ipamorelin, AOD-9604, GHK-Cu (injectable), Melanotan II, KPV, Selank, and Semax.
There are legal risks to using peptides that are not on the approved lists in your med spa. Providers are advised to verify that their compounding pharmacies and outsourcing facilities are properly licensed and/or registered (503A or 503B) and only use adequately sourced ingredients.
The use of “research grade” APIs is not acceptable for human use outside a clinical trial and represents a significant safety and compliance risk. "Gray market" or international suppliers offering products "under the table" are also a red flag that should be avoided by all med spas.
It’s critical to verify that the active ingredient is legally permitted for compounding. If you’re unsure about the status of a peptide product, consult with legal counsel.
Weight Loss Therapies and GLP-1 Medications
Demand for GLP-1 medications such as semaglutide and tirzepatide has expanded the role of med spas in pharmaceutical care.
Many med spas now offer:
Medical weight management programs
Injectable GLP-1 therapies
Metabolic health consultations
These medications are complex drugs with a specific risk profile, which increases the importance of proper prescribing oversight, sourcing, storage, and administration.
The Bottom Line
Operating a med spa safely demands assuring the treatments and services offered meet safety and regulatory standards. Commit to sourcing every product from reputable, verified, and compliant suppliers.
Build collaborations with established 503A pharmacies and 503B outsourcers that prioritize quality, safety, and regulatory compliance. Remain informed about changing regulations and consult with legal and compliance experts when in doubt to protect both your patients and practice.
Scope of Practice: Who Can Provide Med Spa Services?
Medical spa procedures are delivered by multiple healthcare professionals, but each state defines the scope of practice and supervision requirements for each role.
Typical provider hierarchy includes:
Physician (MD or DO)
Nurse practitioner (NP) or physician assistant (PA)
Registered nurse (RN)
Licensed practical nurse (LPN)
Aesthetician or cosmetologist
Delegation
Delegation rules when a provider’s authority is limited vary widely by state.
In reality, delegation practices are being stretched to their legal limits in many med spas, raising patient safety concerns. Delegating procedures involving prescription products, such as compounded sterile preparations, to staff can diffuse accountability and make it more difficult to identify unsafe practices.
Having clear, enforceable supervision standards for med spas that store, administer, dispense, distribute, or otherwise use prescription medication related to their services supports patient safety and helps maintain public trust.
Nurse Practitioner Practice Authority
Across the United States:
Several states allow full practice authority for nurse practitioners (NPs)
Some states require collaborative agreements with physicians
Others require supervised practice periods before independence
These variations affect how medical spas structure clinical oversight.
Corporate Practice of Medicine and Ownership Rules
A key legal doctrine affecting medical spa ownership is the Corporate Practice of Medicine (CPOM).
CPOM laws are designed to ensure that medical decisions remain under the authority of licensed physicians rather than corporations or non-medical business owners.
States generally fall into two categories.
Non-CPOM States
Non-physicians may own healthcare practices, though clinical decision-making must remain under licensed providers.
This means that nurse practitioners (NPs) or physician assistants (PAs) can also own a medical spa in these states.
Examples include:
Florida
Arizona
Washington
Yet, it's critical to understand the specific laws in your state to be certain if NPs and PAs are permitted to own a medical aesthetics practice.
CPOM States
Non-physicians cannot own medical practices.
Examples include:
California
Texas
Illinois
New York
However, some states require only partial physician ownership. For example, California requires physicians to own at least 51%, while an enumerated list of other medical licenses may own up to 49%. Other states restrict medical practice ownership strictly to physicians.
If you live in a CPOM state and do not have a medical license, you cannot own a medical practice. However, in every state (including CPOM states), non-physician entrepreneurs can legally work with medical spas through a legal structure called the MSO.
The Management Services Organization (MSO) Model
In CPOM states, non-physicians often participate in medical spa operations through a Management Services Organization (MSO).
Under this model:
A physician owns the medical practice entity
A separate MSO provides administrative services
Typical MSO services include:
Marketing and branding
Staffing and payroll
Real estate management
Billing and collections
Operational support
These arrangements must be structured carefully to avoid regulatory concerns such as fee-splitting or improper influence over medical decisions.
State Ownership Requirements
SOPs and Protocols for Med Spa Compliance
SOPs (Standard Operating Procedures) and protocols formalize your med spa’s operations. They ensure consistent service delivery, reduce risk, and demonstrate compliance to regulators, auditors, and patients.
Medical treatments or procedures offered in a medical spa typically should have detailed written protocols or standard operating procedures (SOP) developed by the medical director or responsible physician (or NP/PA, where applicable).
Protocols should provide detailed procedures to be followed in administering the treatments and guidance on addressing and responding to adverse incidents. Protocols should be specific enough so that medical decisions are not left to the discretion of the non-physician provider.
Key Components to Include:
Clinical Procedures/Treatment Protocols
Injection protocols, laser treatments, IV therapy administration
Patient assessment checklists and Good Faith Exam documentation
Medication Management
Vendor qualification for 503A/503B compounds and FDA-approved drugs
Proper storage, refrigeration, and inventory controls
Handling of medications
Expiration date tracking and recall procedures
Implementation of applicable USP standards (<795>, <797>, <800>)
Staffing and Delegation
Scope-of-practice documentation for physicians, NPs, PAs, RNs, aestheticians
Supervision and escalation protocols
Safety and Infection Control
Safety, cleaning and disinfection plans and procedures
OSHA compliance, sharps disposal, and PPE protocols
Emergency Preparedness
Response for adverse drug reactions or procedure complications
Emergency contacts, evacuation, and incident reporting procedures
Patient Communication and Documentation
Informed consent forms for procedures and medications
Intake forms
Privacy and HIPAA compliance
Review and complaint handling SOPs
Best Practices:
Keep SOPs updated to reflect changing state laws, new treatments, and new products.
Train all staff regularly and document attendance.
Conduct mock audits or inspections to test adherence.
Maintain a central, accessible repository (digital or physical) for all SOPs.
The Good Faith Exam (GFE)
Many states require a Good Faith Examination (GFE) before medical spa treatments are performed.
This evaluation establishes a legitimate provider-patient relationship and determines whether treatment is appropriate.
This initial medical evaluation determines:
Patient eligibility for treatment
Medical history and contraindications
Appropriate treatment plan
GFEs are typically performed by:
Physicians
Nurse practitioners
Physician assistants
In many states, registered nurses cannot independently perform a GFE because doing so may constitute the practice of medicine.
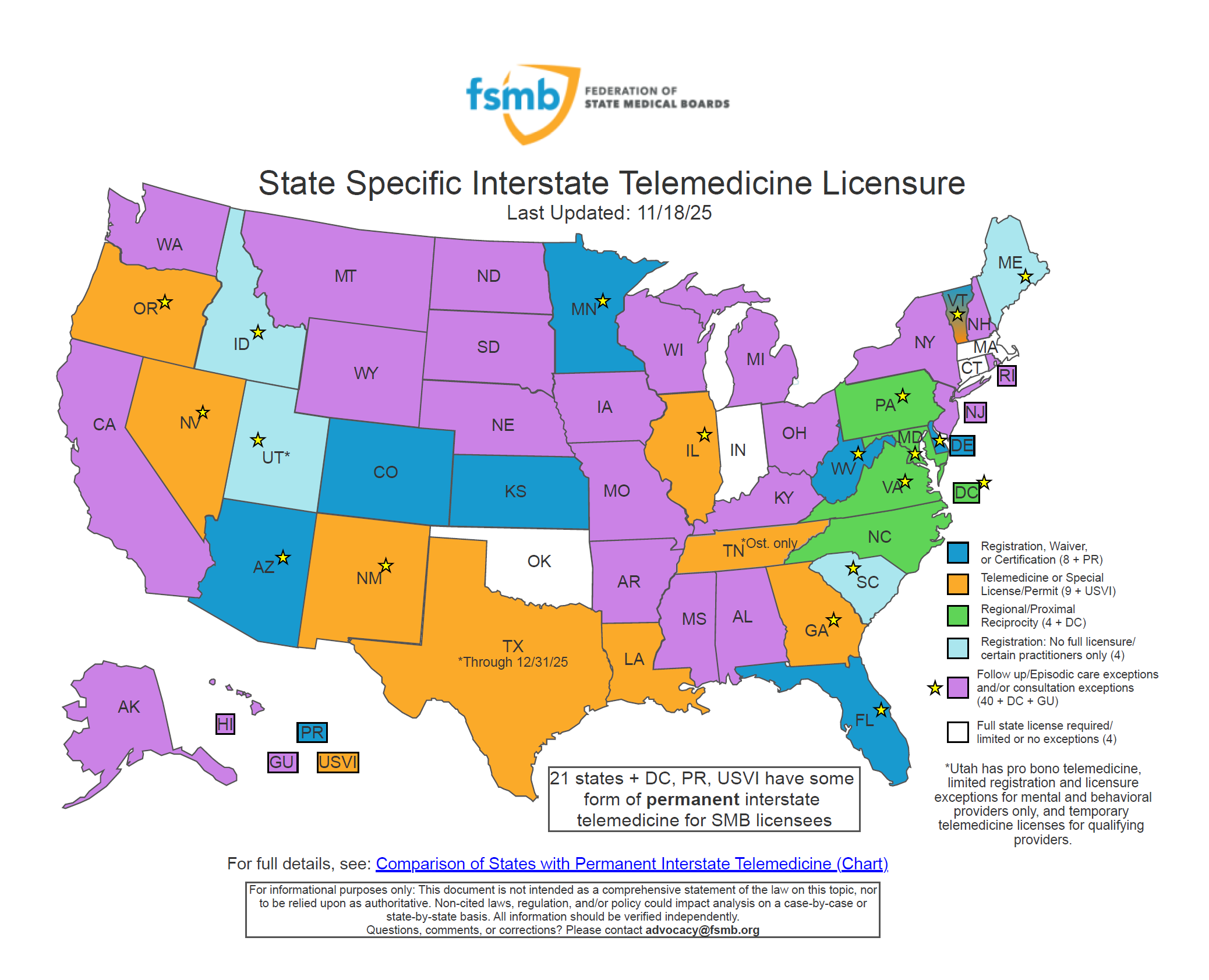
Telehealth and Medical Spas
Telemedicine is increasingly used to conduct Good Faith Exams (GFEs) and consultations in med spas.
While telehealth surged during the COVID-19 pandemic, it’s now becoming more important than ever as patient demand for virtual and digital health services increases. According to a recent PwC survey, 40–42% of Gen Z & Millennials used virtual visits in the last year.
However, telehealth regulations vary widely across states. Providers must ensure compliance with:
State medical licensure rules
Telemedicine practice standards
HIPAA privacy regulations
Clinical documentation requirements
Some states permit telehealth GFEs for aesthetic treatments, while others require in-person evaluations.
While telemedicine is a cost-effective alternative to the more traditional face-to-face method of providing medical care, and one that increasingly meets the needs of younger patients, it’s being cautiously applied in some states. Mainly, it’s important to use telemedicine to actually diagnose and provide care to patients in a compliant and responsible manner.
In Oklahoma, the state medical board passed telemedicine rules exempting physicians from face-to-face meetings with patients if certain criteria are met.
When telehealth is used, it is considered to be rendered at the physical location of the patient, and therefore a provider typically needs to be licensed in the patient’s state. A majority of states have licenses or telehealth-specific exceptions that allow an out-of-state provider to deliver services via telemedicine (called cross-state licensing). It’s important to familiarize yourself with the telehealth licensing requirements for each state (CCHP).
Med Spas and Compounded Medications
Some medications used in medical spas are compounded rather than conventionally manufactured commercially-available FDA-approved products.
Examples may include:
Customized IV hydration mixtures
Vitamin injections
Topical combination numbing creams
Tailored hormone therapies
Compounded weight-loss medications
Sourcing and quality assurance are important considerations for compounded medications.
503A vs 503B vs FDA-Approved Medication Sourcing & Regulations
Med spas may obtain medications through several different regulatory pathways.
503A pharmacies compound medications based on individual patient prescriptions.
503B outsourcing facilities were established under the Drug Quality and Security Act (DQSA) to allow outsourcing facilities to produce compounded sterile medications for healthcare facilities.
Med spas may also obtain medications from 503A compounding pharmacies, which:
Compound medications based on patient-specific prescriptions
Operate primarily under state board of pharmacy oversight
Unlike 503B facilities, 503A pharmacies generally cannot supply medications in bulk for office use.
Because of this limitation, many med spas rely more heavily on 503B outsourcing facilities.
Many medical spas purchase compounded medications from 503B outsourcing facilities.
503B outsourcing facilities may:
Compound sterile medications in bulk
Supply drugs to healthcare facilities for office or “office-administered” use
Produce medications without patient-specific prescriptions
This model allows med spas to purchase compounded sterile injectables in advance rather than obtaining individual prescriptions.
However, med spas must verify that suppliers are:
Registered with the FDA and regularly inspected
Subject to cGMP compliance
Producing drugs in accordance with FDA requirements, including applicable drug shortage provisions where relevant
Offering transparent testing data and batch documentation
Supplier Due Diligence and Medication Safety
Medical spas should implement strong protocols for medication sourcing and verification.
Key compliance measures include:
Product Verification
Confirm suppliers are licensed and authorized
Verify FDA registration of outsourcing facilities
Review certificates of analysis where available
Storage and Handling
Monitor temperature conditions
Maintain controlled storage (depending on specific medication)
Track expiration or beyond use dates and lot numbers
Documentation
Maintain detailed records of:
Product receipt
Storage conditions
Patient administration
Lot and batch numbers
Adverse Event Reporting
If adverse reactions occur, providers may need to report events through FDA MedWatch.
Patient Safety and Enforcement Actions
Regulatory enforcement actions involving compounded medications in aesthetic clinics have highlighted potential risks related to:
Improper sterile preparation
Counterfeit medications
Repackaging under non-sterile conditions
Examples include:
A Tennessee case involving counterfeit weight-loss drugs produced in a home laboratory
Sterility failures involving compounded tirzepatide products
Repackaging of injectable medications under non-sterile conditions
These incidents have increased scrutiny on medication sourcing and clinical oversight in med spa environments. They underscore the importance of sourcing medications only from legitimate, regulated suppliers.
Top 10 Compliance Mistakes Med Spas Make
Even experienced operators may inadvertently overlook key regulatory requirements. The most common compliance issues observed in medical spa environments include:
1. Lack of a documented Good Faith Exam
Treatments performed without a properly documented and conducted initial medical evaluation may constitute the unauthorized practice of medicine.
2. Improper delegation of medical procedures
Certain treatments are performed by staff who are not authorized under state scope-of-practice laws.
3. Noncompliant ownership structures
Violations of corporate practice of medicine laws occur when non-physician owners exert control or influence over medical decisions. Even if all providers are properly licensed, allowing business owners without medical credentials to dictate treatments, procedures, or clinical protocols creates legal risk and undermines patient safety.
4. Purchasing compounded medications from unverified suppliers
Failure to verify compounding pharmacy credentials creates safety and legal risks.
5. Improper drug storage
Failure to maintain appropriate temperature controls, storage conditions, and inventory monitoring can compromise medication integrity, create regulatory violations, and harm patient safety.
6. Inadequate documentation of treatments
Incomplete or inconsistent patient records can create serious regulatory and legal exposure. This includes missing informed consent forms, lack of GFE documentation, incomplete procedure notes, inaccurate medication administration logs, or failure to track lot numbers and expiration dates of injectable products.
7. Improper telehealth evaluations
Virtual consultations may not meet state standards for establishing a provider-patient relationship. Virtual consultations that do not adhere to state-specific telemedicine requirements can create compliance risks.
8. Lack of written protocols and SOPs
Many med spas operate without documented clinical procedures.
9. Misleading advertising claims
Marketing language may imply FDA approval, guaranteed results, or non-evidence-based claims.
10. Lack of compliance oversight
Many medical spas do not designate a compliance officer or implement internal audits. Without consistent oversight and a clear strategy, many compliance gaps can emerge.
States with the Most Active Med Spa Regulatory Oversight
While every state regulates healthcare providers, some states have taken a more active approach toward med spa oversight.
States frequently considered among the most closely regulated include:
California
Strict corporate practice of medicine laws and strong medical board enforcement. Requires a physician or physician-owned corporation with at least 51% doctor ownership. Non-physicians join via MSOs. The medical director must be a physician and stay actively involved.
Texas
Detailed guidance from the medical board on delegation and supervision. Physician-only owned. MSOs can be used to get non-physicians involved. The medical director stays hands-on for all procedures.
New York
Strict ownership restrictions and active enforcement. Only physicians can own med spas via professional entities, while MSOs bring in non-physicians. The doctor owner is also the medical director and must be hands-on.
Ohio
Pharmacy oversight through terminal distributor licensing. While anyone can own a med spa if they hire a physician as medical director, non-physicians generally cannot directly receive compensation tied to the provision of medical services.
Florida
Rapid med spa growth has prompted increased regulatory attention. Non-physicians can own med spas as long as they don't control the medical affairs. The medical director must be a licensed physician.
Illinois
Active enforcement related to corporate practice of medicine and supervision rules. CPOM is enforced, so physicians must own med spas. APRNs with full practice authority can also take part. The medical director can be a physician or an APRN with authority.
Because state laws evolve frequently, med spa operators should review current guidance from state medical boards, pharmacy boards, and regulatory agencies.
Essential Elements of a Med Spa Compliance Program
A strong medical spa compliance program typically includes the following components.
Licensing and Credentialing
Ensure that:
All providers maintain active state licenses
Medical directors meet supervisory requirements
Facility permits are obtained where required
HIPAA Compliance
Med spas must comply with federal patient privacy regulations and protect patient health information through:
Secure electronic health record systems
Controlled staff access to patient data
Privacy policies and breach protocols
OSHA and Workplace Safety
Compliance with occupational safety standards includes:
Safe handling of chemicals and biologics
Medical waste disposal procedures
Infection control protocols
Equipment maintenance
Informed Consent
Before treatments, patients must receive:
Clear explanation of risks, benefits, and alternatives
Disclosure of off-label or compounded therapies
Written informed consent documentation
Marketing and Advertising Compliance
Medical spa media, marketing, and advertising must remain accurate, truthful, and non-misleading. As with all medical practices, medical spas and aesthetic practices must conform to state and medical board advertising rules.
Key considerations include:
Avoiding exaggerated outcome claims
Clearly identifying licensed providers
Clearly and accurately listing practitioner and provider qualifications, credentials and experience
Avoiding statements implying FDA approval where none exists
Statements on websites, including testimonials from patients, must be objectively provable as accurate and true. This applies to all public-facing content, including patient photos on social media and websites.
In addition, posts and interactions in social media must preserve and protect patient confidentiality and HIPAA protections. Patient photos should only be used with the express written permission of the patient, in full compliance with federal and state privacy laws. Reasonable steps should be taken to protect the patient’s identity.
Ideally, all advertising, marketing, and other public statements be reviewed by knowledgeable legal counsel.
Building a Compliance-Ready Med Spa
To wrap up, medical spas that prioritize compliance tend to implement structured operational systems such as:
Standard operating procedures
Staff regulatory training programs
Internal compliance audits
Detailed patient documentation protocols
Regular compliance reviews often examine:
Treatment documentation
Informed consent forms
Provider licenses
Medication inventory records
Med Spa Compliance Consulting
As regulatory oversight of medical spas continues to evolve, many operators seek specialized guidance to navigate the complex intersection of medical, pharmacy, and business regulations.
Compliance challenges commonly arise when medical spas expand services involving:
Injectable medications
IV hydration therapy
GLP-1 weight-management treatments
Compounded sterile products
Telemedicine consultations
Building a sustainable medical spa operation requires careful alignment between clinical oversight, medication sourcing and storage, facility operations, and state regulatory requirements.
Restore Health Consulting works with healthcare entrepreneurs, med spas, and regulated wellness centers to design compliance frameworks for emerging healthcare models.
These engagements may include support for:
Med spa regulatory assessments and licensure
Compounded medication sourcing and drug storage strategies
Compliance program development
Build a Scalable and Compliant Med Spa Business from Day One
Ensuring med spa compliance comes down to your state, operations, and business model. Regulations are nuanced and fast-changing, but you should always focus on building a strong compliance core from day one.
Don’t let regulatory uncertainty slow down your med spa launch or expansion. Restore Health Consulting can help every step of the way, whether you’re in the planning stage or already have a med spa up and running but need tailored advisement.
We can help support your medical, pharmacy, and operational compliance needs, allowing you to focus on building a scalable medical spa model while maintaining strong patient safety and regulatory alignment.
Ultimately, a compliance-driven approach not only protects your patients but also strengthens the credibility and sustainability of the medical spa industry.
References:
1. National Association of Boards of Pharmacy (NABP). Inside the Fight Against Falsified Medications: How Traceability Protects Patients; Policy Perspectives Injecting Clarity: The State of Medical Spa Regulation in the US. https://nabp.pharmacy/. Published February 2026. Accessed March 6, 2026. https://www.nxtbook.com/nabp/innovations/innovations-magazine-jan-feb-2026/index.php#/p/1
2. Laws for Opening a Med Spa. American Med Spa Association. https://americanmedspa.org/opening-a-med-spa-laws
3. PricewaterhouseCoopers. US Healthcare Consumer Insights and Engagement Survey: PwC. PwC. Published 2024. https://www.pwc.com/us/en/industries/health-industries/library/healthcare-consumer-insights-survey.html
4. FSMB | FSMB Policies on Telemedicine / License Portability. Fsmb.org. Published May 2, 2024. https://www.fsmb.org/advocacy/telemedicine-policies/
5. State Telehealth Policies for Cross-State Licensing. CCHP. https://www.cchpca.org/topic/cross-state-licensing-professional-requirements/
6. Counterfeit Version of Botox Found in Multiple States. FDA. Published online 2024. https://www.fda.gov/drugs/drug-safety-and-availability/counterfeit-version-botox-found-multiple-states
7. Wang JV, Hattier G, Rohrer T, Zachary CB, Saedi N. Experiences With Counterfeit Aesthetic Medical Devices and Injectables: A National Survey. Dermatologic Surgery. 2019;46(10):1323-1326. doi:https://doi.org/10.1097/dss.0000000000002307
8. Interim Policy on Compounding Using Bulk Drug Substances Under Section 503A of the Federal Food, Drug, and Cosmetic Act. U.S. Food and Drug Administration. Published 2025. https://www.fda.gov/regulatory-information/search-fda-guidance-documents/interim-policy-compounding-using-bulk-drug-substances-under-section-503a-federal-food-drug-and
9. THE AMERICAN MED SPA ASSOCIATION. GUIDELINES for NON-INVASIVE MEDICAL AESTHETIC PRACTICES (MEDICAL SPAS) PREPARED BY.; 2020. https://facialesthetics.org/wp-content/uploads/2020/10/amspa_practice_guidelines.pdf